Article
Counting Spermatozoa - Part 2 Return to to part 1
By B. W. Pickett, Ph.D.
Professor Emeritus, Colorado State University
In Part 1 five factors were listed that significantly influence the use of a hemacytometer to count the number of spermatozoa in a semen sample. They are as follows:
a) The number of chambers that must be counted for reasonable accuracy.
Figure 1. Hemacytometer (black arrow), dilution media
(Unopette, white arrow) and high quality, phase-contrast
microscope used to determine concentration of spermatozoa.
Considering all the variables associated with hemacytometer counts, it was statistically determined that a minimum of eight chambers should be counted to determine a mean accurate count for a single sample of semen. In the event any one of the eight counts is 10% or more above or below the mean, that count should be eliminated and another count made. Under these circumstances, the accuracy of the hemacytometer is superior, in general, to an electronic method, but the precision (or deviation) is less with the electronic counter, because there are fewer variables in preparing the semen sample for counting. Further, the human (technician) error involved in preparing a sample for electronically counting is relatively minimal compared to preparing a sample for counting in the hemacytometer.
2b) The time required to prepare a sample for counting with the hemacytometer.
Figure 2. Puncturing Unopette reservoir using
the pointed shield. In recent years a system for
counting cells with a hemacytometer (Unopette)
has been developed (Becton-Dickinson, Rutherford,
NJ) that has certainly simplified the older methods,
which utilized a white or red blood cell pipette.
In recent years a system for counting cells with a hemacytometer (Unopette) has been developed (Becton-Dickinson, Rutherford, NJ) that has certainly simplified the older methods, which utilized a white or red blood cell pipette.
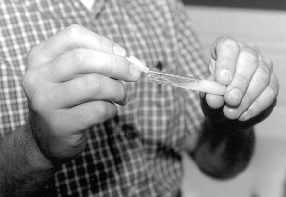
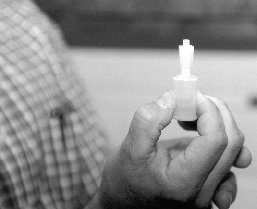
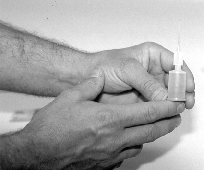
Presented in Figure 1 is the equipment necessary for utilizing this newer method of counting spermatozoa. Once the semen sample has been collected and thoroughly mixed, a sub-sample is taken for ease of sampling. The Unopette is used to puncture the plug of the reservoir with the pointed end of the plastic shield (Figure 2). With the shield in place, and serving as a cap for the reservoir, remove the capillary tube (Figure 3) Submerge the capillary tube in a thoroughly mixed sample of raw semen (Figure 4). Allow semen to fill the capillary tube, then remove it from the sample. Wipe excess semen from the outside of the capillary tube, being careful not to remove any semen from within the capillary tube. Remove cap and lightly squeeze the reservoir between thumb and forefinger. Place pipette into the reservoir and release the pressure (Figure 5), this allows semen to be siphoned into the reservoir. Rinse the capillary tube by gently squeezing the reservoir several times without forcing fluid out of the top. Remove and invert the pipette (Figures 6 and 7).
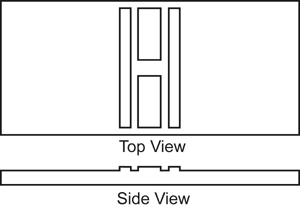
Presented in Figure 8 is a schematic drawing of the top and side view of the hemacytometer. There is a grid on each of the two shorter, wider portions, (chambers) of the hemacytometer. The two elongated portions (one on each side of the chambers) are slightly higher than the chambers. Consequently, when the weighted cover slip is placed over the chambers, there is a very specific volume of diluted semen over the grid, which must be used as one of the multiplication factors to calculate the number of spermatozoa per milliliter of semen.
Figure 8. Drawing of the top and side view of a hemacytometer.
c) The time required to count the number of spermatozoa in the appropriate
number of squares.
Obviously, the length of time required to prepare and count the sample (including the calculations) will vary from 10 to 20 minutes for the two chambers (one on each side) depending upon the experience of the technician. It must be emphasized that a phase-contrast microscope must be used for counting. The phase-contrast microscope, when properly cleaned and adjusted, permits clear observation of individual particles, cells, spermatozoa, etc., of similar optical density, without the necessity of staining, unseen transparent cells can be seen in rich contrast to their background. Consequently, without a phase-contrast microscope it is difficult, if not impossible to differentiate some cellular debris from spermatozoa.
d) Procedure for counting the spermatozoa.
Place the hemacytometer on a flat surface, such as a counter top. Position the special weighted cover slip so that it is centered and cover both counting grids (Figure 9). Load the diluted sample onto the hemacytometer by allowing a single drop of diluted semen to be drawn across the counting grid by capillary action (Figure 10). To assure an accurate count, do not overfill the space between the cover slip and the counting grid. If overfilling occurs, the chamber must be dismantled, cleaned and the procedure repeated. Load both counting chambers on the hemacytometer. Place the loaded hemacytometer on the microscope stage (Figure 11). Locate the counting grid on the monitor. Adjust magnification so that the central block of 25 squares can be observed (Figure 12). Count the spermatozoa in the 25
squares on both sides of the hemacytometer. Be sure to count all spermatozoa within a square and only those spermatozoa on or across two of the four borders. This will prevent counting the same spermatozoon twice on two adjoining squares. At a 1:100 dilution, this total number represents millions of spermatozoa ml in this sample of gel-free semen. For a reasonably accurate count, two chambers (25 squares/ chamber) should be counted on four hemacytometers, thus duplicate counts made on each sample of semen. Since utilization of a hemacytometer for estimating sperm numbers can be very time consuming, more rapid procedures would save time, and thus, eventually money.
Counting is further complicated when semen samples are very dilute or extremely thick, which changes the number of large squares that must be counted for sufficient accuracy.
e) Preparing and counting a sample of semen requires considerable
technical skill and equipment. Therefore, very few farms are prepared
to make accurate counts with a hemacytometer.
It should be noted that the hemacytometer requires some special care. A dirty or scratched instrument will result in inaccurate counts. The following steps should be done immediately after use.
1. Wash the hemacytometer chamber and cover slip individually with a
mild detergent (Ivory). Be careful not to scratch the chamber. Rub the
surface gently with a facial tissue, lens paper or fingertip.
2. Rinse well in warm water.
3. Rinse in distilled or deionized water.
4. Rinse in alcohol or acetone to dry or dry with lens paper.
5. Store in a dust-free location with the cover slip and hemacytometer
wrapped separately in tissue.
About the Author...
B. W. Pickett, Ph.D is Professor Emeritus, Colorado State University.


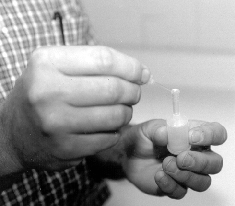
Figure 3. Removing capillary tube from the pointed shield, which will be used to load an appropriate volume of raw semen for an accurate hemacytometer count.
Figure 4. Loading an appropriate volume of raw semen by capillary action for counting in a hemacytometer.
Figure 5. Adding raw semen to diluent reservoir in order to provide proper dilution for an accurate hemacytometer count.
Figure 6. Removing pipette from diluent reservoir
Figure 7. Inverting pipette and placing it on the
reservoir to be used as a dispenser in loading
the hemacytometer.
Figure 9. Placing weighted cover slip over both etched counting surfaces of the hemacytometer.
Figure 10. Loading the hemacytometer by placing a single drop of diluted semen in the loading slot of the hemacytometer.
Figure 11. Placing loaded hemacytometer on the stage of a phase-contrast microscope so that the counting grid is visible on the monitor.
Figure 12. A drawing of the 25 squares to be counted on a grid of a loaded hemacytometer..
Animal Reproduction Systems
800-300-5143